Assessment of Persistent Chemicals of Concern in White Sturgeon (Acipenser transmontanus) in the Hanford Reach of the Columbia River, Southeastern Washington, 2009
Links
- Document: Report (1.6 MB pdf) , HTML , XML
- Download citation as: RIS | Dublin Core
Acknowledgments
Several individuals and organizations contributed to this study. We give special thanks to Brett Tiller, Environmental Assessment Services, LLC, for providing us with the samples and Laura Buelow, U.S. Environmental Protection Agency, Region 10, for her invaluable and helpful discussions. We also would like to thank Steve Zaugg, U.S. Geological Survey (deceased) for laboratory support and assistance.
Abstract
White sturgeon (Acipenser transmontanus) are long-lived, late-maturing, benthic-feeding fish that are ideal candidates for assessing the bioaccumulation of persistent chemicals. In this study, composite tissue samples of brain, liver, gonad, and fillet were collected from white sturgeon in 2009 from five sites in the Hanford Reach of the Columbia River near Hanford, Washington. The composite tissue samples at each site were analyzed for the concentrations of individual chemicals as well as the total concentrations of four chemical classes: (1) organochlorine (OC) pesticides, (2) industrial or personal care products, (3) polybrominated diphenyl ether (PBDE) congeners, and (4) polychlorinated biphenyl (PCB) congeners. The results showed that chemicals from all four classes were present in the fish, and that OC pesticides and degradation products (such as oxychlordane, fipronil sulfide, and dichlorodiphenyltrichloroethane (DDT) degradates, PBDE congeners, and PCB congeners) often were present in all tissues and at all sites. Gonad tissues generally had the highest total concentration of each chemical class, followed by brains, livers, and fillets. The concentrations of several chemicals or chemical classes exceeded many of the human health benchmarks for two different populations (general/recreational consumers and subsistence/Tribal consumers), and this was especially true for the total concentrations of DDT degradation products and PCB congeners. These results suggest that continued monitoring of resident fish in the Hanford Reach, as well as assessments of the health impacts on consumers of those fish, are warranted.
Introduction
The Columbia River (fig. 1) is an invaluable resource in the Pacific Northwest, known for its scenery, recreational opportunities, and specialty agriculture, and as a major source of hydroelectric power for the region. Originating in the Canadian Rocky Mountains, the Columbia River traverses 2,000 kilometers (km) (1,243 miles [mi]) through the Pacific Northwest, draining a 669,550-square kilometer (km2) (258,513 square mile [mi2]) area, and discharges into the Pacific Ocean. Fish populations within the Columbia River have declined since the late 1880s and contributing to these declines are several stressors that include dams, overharvest, habitat loss, invasive species, predation, and, more recently (1960s), anthropogenically derived chemicals. The accumulation of toxic chemicals can threaten the survival of fish species and can harm other non-fish species that consume contaminated fish, such as birds and mammals (Herger and others, 2017). Additionally, Columbia River fisheries are a major source of food for members of the Columbia Basin Treaty Tribes (Columbia River Inter-Tribal Fish Commission [CRITFC], 1994), and studies have shown that tissues of resident fish in the Columbia River can contain high concentrations of contaminants, such as organochlorine (OC) pesticides (including dichlorodiphenyltrichloroethane [DDT] and its degradation products), polychlorinated biphenyls (PCBs), polybrominated diphenyl ethers (PBDEs), and metals (U.S. Environmental Protection Agency [EPA], 2002, 2009; Oregon Department of Environmental Quality [ODEQ], 2012; Washington State Department of Health [WSDOH], 2012, 2017; Herger and others, 2017). Tribal members and those non-Tribal subsistence fishers who consume fish at higher rates may have a direct pathway of exposure to persistent toxic chemicals (Rifkin and LaKind, 1991, CRITFC 1994; EPA, 2002) via the consumption of resident fish from the Columbia River.
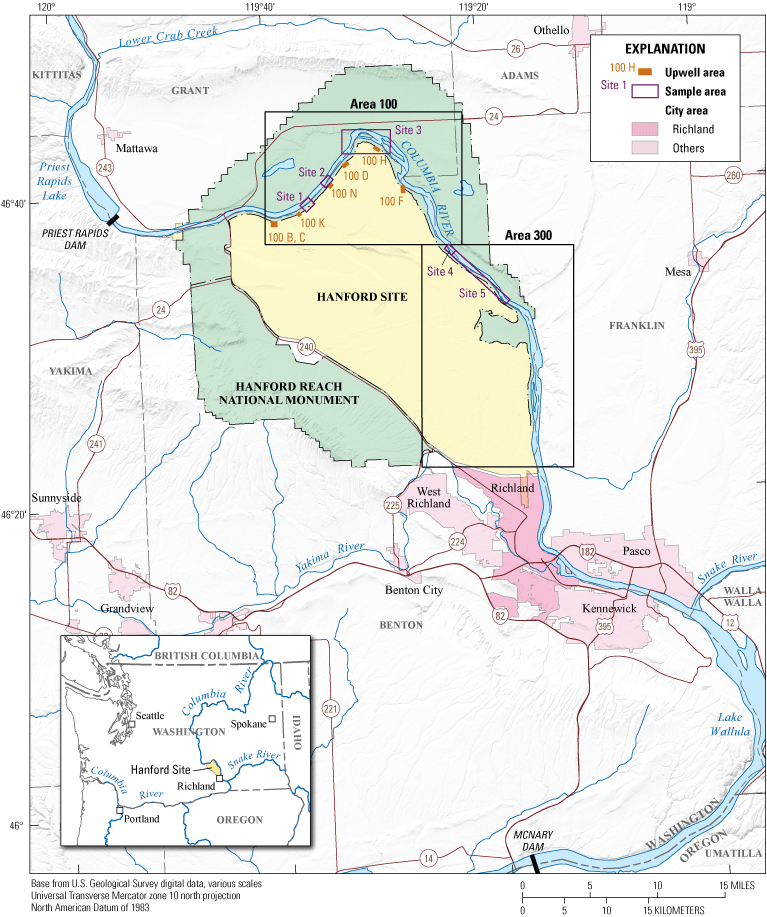
The Hanford Reach of the Columbia River near Hanford, Washington, and sites where white sturgeon (Acipenser transmontanus) were sampled for persistent chemicals of concern. Some sampling locations at sites 4 and 5 converge at the boundary between those sites.
White sturgeon (Acipenser transmontanus) inhabit several large river basins along the Pacific Coast of North America. In the Columbia River Basin, white sturgeon historically moved freely and ranged from the Pacific Ocean to upstream approximately 1,300 km (808 mi) into Canada (North and others, 1993) and in the Snake River up to Shoshone Falls near the City of Twin Falls, Idaho. Construction of hydroelectric dams beginning in the 1930s on the Columbia River resulted in the segregation of white sturgeon into discrete populations (Beamesderfer and others, 1995), with their movements and distribution greatly restricted to specific reservoirs. North and others (1993) noted that recaptured tagged sturgeon traveled an average distance of 8.1 km (5 mi) from the original tagging location. Furthermore, a radio telemetry study of white sturgeon in 2002 and 2003 in the lower Columbia River between river km 46 and 52 documented strong site fidelity after dispersal during periods of seasonal migration (Parsley and others, 2008). In 2000, researchers using acoustic telemetry to document movements of white sturgeon in The Dalles Reservoir during the spawning season showed that the fish generally did not move great distances (less than 1 km [0.6 mi] during the monitoring period; Ward, 2002). Although studies have shown that white sturgeon are apt to remain in relatively localized areas (Haynes and others, 1978; Ward, 2002; Hildebrand and others, 2016), some sturgeon move considerable distances related to seasonal changes and spawning. Haynes and others (1978) documented that tagged sturgeon in the mid-Columbia River between McNary and Priest Rapid Dams began their movements in mid-June and ceased movements in mid-October, and that movements during the months of November through May were limited and short in duration. Considered the largest freshwater fish in North America, white sturgeon also use estuarine environments, although marine movements between river basins are infrequent (Hildebrand and others, 2016). Therefore, Columbia River white sturgeon spend most of their life cycle in the Columbia River and its tributaries and can be a useful indicator of the local exposure to contamination and, therefore, a way to indicate local sources of contaminants.
Bioaccumulation occurs when an organism accumulates a substance at a rate faster than it can eliminate that substance. White sturgeon are a long-lived, late-maturing, benthic species, making them particularly susceptible to contaminant accumulation because hydrophobic contaminants such as OC pesticides and PCBs accumulate in bed sediments (Devore and others, 1995; Feist and others, 2005). Bottom-dwelling aquatic species like white sturgeon also are vulnerable to contaminant exposure indirectly through the food web or from suspended contaminated sediments (Seelye and others, 1982). Many of these chemicals have been detected previously in multiple species of fish and sediments in the lower, mid-, and upper Columbia River (Foster and others, 2001; Pacific Northwest National Laboratory [PNNL], 2005; Hulstrom, 2011; WSDOH, 2012, 2017; Nilsen and others, 2014). Additionally, previous studies have shown that sediments in the Columbia River contain sediment-sorbed pollutants (Buck, 2004; Hayslip and others, 2007; Counihan and others, 2014; Nilsen and others, 2014), allowing sturgeon and other species to ingest sediment derived pollutants through the food web. The sturgeon’s longlife span and benthic lifestyle increase the opportunity for exposure and accumulation of these chemicals. For these reasons, white sturgeon that have incurred bioaccumulation of chemicals in their tissues, along with their strong site fidelity and constrained movements, can be useful indicators of local exposure to chemical contamination.
Effects of chemicals such as OC pesticides, PCBs, and PBDEs bioaccumulating in fish include endocrine disruption, poor reproductive success, feminization, and neurotoxicity (Feist and others, 2005; Wan and others, 2007; Brar and others, 2010, Gundersen and others, 2017; Herger and others, 2017). Focusing studies on tissue distribution of chemical bioaccumulation will increase our knowledge of the pathways these chemicals take after exposure. White sturgeon take several years to reach full sexual maturity (Foster and others, 2001), and populations in impounded stretches of the Columbia River are lacking reproductive success (Beamesderfer and others, 1995). Although there are several factors responsible for the adverse health effects of these fish, exposure to chemicals could be a contributing influence. With increasing knowledge of tissue distribution, future studies can focus on the health effects that chemical bioaccumulation can have on white sturgeon and other species of fish by knowing organ-specific disruption through chemical exposure. Chemicals considered to be endocrine disruptors and carcinogenic, such as OC pesticides and PCB congeners, have been detected in sampled white sturgeon in the Columbia River Basin (EPA, 2002; Feist and others, 2005; Herger and others, 2017). In studies of Russian and Caspian Sea sturgeon species, accumulations of heavy metals and pesticides were detected in gonads, livers, and muscles (Khodorevskaya and others, 1997; Kajiwara and others, 2003), indicating that similar contamination threats are worldwide. Other studies on Columbia River white sturgeon suggest that exposure to chemicals, including OC pesticides and PCBs, may be affecting growth and reproduction (Foster and others, 2001; Feist and others, 2005); however, these studies focused on liver and gonad tissues. Additionally, studies of Caspian Sea and Siberian sturgeon species also have detected abnormalities in gonad development, nonviable embryos, and reproductive anomalies resulting from chemical accumulation (Khodorevskaya and others, 1997; Akimova and Ruban, 2009). Tissue-focused studies could increase insights into the monitoring of long-term accumulation of chemicals, maternal transfer, and age-related trends of contamination (Peng and others, 2012; Dang and others, 2016). Moreover, understanding tissue distribution of chemicals is crucial from a human health standpoint, as consumers of fish may consume more than one type of tissue, this is especially true for Tribal/subsistence fisher consumers (CRITFC, 1994). Further studies to assess the relation of anthropogenic chemicals to negative health impacts on white sturgeon would help inform decisions about fish consumption. Those studies can be compared with studies of contaminant accumulation in other sturgeon species to develop a full understanding of the contaminants’ effects. Continued monitoring of contaminated tissues that may be used for consumption also is warranted.
Study Area
The Hanford Reach is an unimpounded 81 km (50 mi) stretch of the Columbia River upstream from McNary Dam in southeastern Washington that ranges from approximately river km 628 to 547 (fig. 1). The Hanford Site, a 1,517 km2 (586 mi2) Federal facility situated alongside the Hanford Reach, was originally established as part of the Manhattan Project (Gosling, 2010). The Hanford Site is of interest because of the legacy from its past operations, which included liquid waste disposal that contributed to the contamination of local groundwater that ultimately reached the Columbia River. A plutonium production facility operated at the site from 1944 to 1988. As a result of these activities, the Hanford Site had several known and unknown waste disposal locations that included subsurface burial sites, liquid waste disposal sites, and contaminated facilities. Radioactive, nonradioactive, mixed, and hazardous waste was produced at the Hanford Site during its time of operation (PNNL, 2000), and several of the hazardous waste disposal locations at the facility contained bioaccumulating chemicals such as lead and mercury, pesticides such as DDT, and other chemicals (including PCBs and dioxin). These waste disposal practices contaminated upland soils and groundwater beneath the site (Hulstrom, 2011). Although operations at the Hanford Site ended in the late 1980s, contamination of the surrounding land and water continues to be a concern, likely due to groundwater movement from the contaminated areas to the Columbia River. Additionally, various other sources are located upstream from the site, such as agricultural runoff, mining operations, paper mill production, atmospheric deposition, runoff from urban land, and permitted wastewater discharges that are potential sources of bioaccumulating chemicals (EPA, 2009; Hulstrom, 2011).
The Hanford Site is divided into operational areas to facilitate security, safety, and functionality (PNNL, 2000); the location of the sampling sites used for this study are within the predetermined sub-areas currently used at the Hanford Site (PNNL, 2000; Hulstrom and Tiller, 2010). The sub-areas at the Hanford Site relevant to this study are shown in figure 1. The sturgeon sampled for this study were collected at five sites within sub-areas 100 and 300. Sub-areas 100 and 300 were chosen for their proximity to the Hanford Site. Approximately 32 km (20 mi) of the Columbia River flows alongside sub-area 100, and this area contained all nine plutonium production reactors, several waste disposal sites, barrel landfills, contaminated soil containing aldrin, dieldrin, DDT, paint waste, petroleum waste, and sodium dichromate (EPA, 1997; Capron, 2008). Sub-area 300 previously housed the fuel manufacturing operations and experimental laboratory facilities and is located alongside the Columbia River upstream from the City of Richland. Sub-area 300 had more than 70 waste burial grounds and unlined surface waste ponds and more than 130 contaminated facilities, all of which are contaminated with uranium, transuranic waste (plutonium), lead, toxic metals, benzene, PCBs, polycyclic aromatic hydrocarbons (PAHs), and various solvents (Peterson and others, 2008; Agency for Toxic Substances and Disease Registry, 2011).
Purpose and Scope
The focus of this study was the bioaccumulation of four classes of persistent chemicals in white sturgeon collected from the Hanford Reach of the Columbia River near the Hanford Site. The four classes consisted of OC pesticides and pesticide degradation products, industrial or personal care products, PBDE congeners, and PCB congeners. Some of the waste from local contaminated groundwater produced by historical operations at the Hanford Site that reached the Columbia River was eventually deposited on the riverbed sediment (PNNL, 2005; Hulstrom, 2011). Although not included in this study, radioactive materials and chemicals were among the contaminants released from the Hanford Site over time, and their occurrence is described elsewhere (PNNL, 2005; Capron, 2008; Hulstrom, 2011). The study described in this report had three objectives: (1) to determine whether persistent chemicals of concern are bioaccumulating in white sturgeon residing in the Hanford Reach, (2) to identify where in the fish the chemicals are accumulating, and (3) to determine whether there are implications for human health related to the bioaccumulation of persistent chemicals of concern in white sturgeon.
The chemicals analyzed in this study consisted of 25 OC pesticides and degradation products (table 1), 6 industrial or personal care products, 10 PBDE congeners, and 15 PCB congeners (table 2). These 56 chemicals (lipid content was not included) were analyzed in brain, muscle, gonad, and liver tissues and, therefore, provided insight into the bioaccumulation that occurs in resident white sturgeon near the Hanford Site. The sampling results from this study were used to determine whether persistent chemicals of concern are bioaccumulating in white sturgeon and to assess the relative amount of bioaccumulation occurring in different tissues within those fish. The results also were used to assess implications for human health by comparing the measured concentrations to available health benchmarks. Because the samples were obtained from five sites and four types of fish tissue and organs, and included 56 chemicals per sample, the raw data contained more than 1,000 discrete measurements. All resulting data were previously published, including metadata for each fish—its sex and weight, as well as its location and date collected (Nilsen and others, 2016).
Table 1.
Organochlorine pesticide chemicals analyzed in samples collected from the Hanford Reach of the Columbia River near Hanford, Washington.[Insecticides: DDD, dichlorodiphenyldichloroethane; DDE, dichlorodiphenyldichloroethylene; DDT, dichlorodiphenyltrichloroethane]
Table 2.
Chemicals analyzed in samples collected from the Hanford Reach of the Columbia River near Hanford, Washington.[Polybrominated diphenyl ether congeners: BDE, brominated diphenyl ether. Polychlorinated biphenyl congeners: chemical is no longer legal for use in the United States. PCB, polychlorinated biphenyl]
Methods
Field Methods
White sturgeon were collected from five locations in the Hanford Reach of the Columbia River during July 16–September 15, 2009 (Hulstrom and Tiller, 2010). Sturgeon tissues from that sampling effort were provided to the U.S. Geological Survey (USGS), and the USGS did not capture the fish. The fish were captured by use of long-line techniques, whereby circle hooks were attached at 4.6 m (15 ft) intervals along a 61–91 m (200–300 ft) rope let out along the river. One hundred sixty-six sturgeon were captured during the collection period, but only 30 were initially retained to comply with State regulations enacted to ensure that mature fish are not removed from the fishery. Fish that did not meet the legal requirements of fork lengths from 110 to 137 centimeters (cm) were released back to the river after recording their physical measurements. After capture and weight measurements were taken, the fish that complied with State regulations were retained and euthanized by severing the spinal cord, and the gills and organs were preserved for analysis. Twelve of these 30 fish had anatomical abnormalities, including duplicate fins, lesions, and discolored gonads. Of the 30 fish retained, tissues to be analyzed from 16 sturgeon were provided to the USGS, and the resulting data (Nilsen and others, 2016) were examined for this study. Sturgeon analyzed in this study had a mean length of 133 cm and a mean weight of 10.4 kilograms (kg). Samples of brain, gonad, and liver tissue and a subsample of the final fillet sample were individually collected from the sturgeon. The samples then were composited, by tissue type and among all the fish at each site location, resulting in one sample of each of the four tissues, from each of the five locations, with masses of 10–20 grams or more, as available. These composited tissue samples were wrapped in aluminum foil, frozen, and shipped to the USGS National Water Quality Laboratory (NWQL) in Lakewood, Colorado, for analysis.
Laboratory Analytical Methods
Sturgeon samples were analyzed in 2011 at the NWQL. Laboratory procedures were based on techniques described in Zaugg and others (2007) and were modified for this study. These modified laboratory procedures were subsequently used and published in Nilsen and others (2014). After receipt at the NWQL, the frozen samples were thawed and homogenized using a blender with specialized glass, stainless steel, and Teflon® internal parts. Extraction of the chemicals of interest from the homogenized samples was completed by using pressurized liquid extraction. The chemicals of interest were isolated from the extract after clean-up by capillary column gas chromatography. The results then were detected by negative ion mass spectrometry using selected ion monitoring. Concentrations of the chemicals of interest were reliably quantified at levels above the method quantitation limit. The qualitative identification of compounds at concentrations less than the method quantitation limit were reported as an estimate. The results for each of the samples were reported in micrograms per kilogram wet weight. All results from this study are available in the associated data release (Nilsen and others, 2016).
Quality Control
The results from the environmental samples were validated with quality assurance measures that included three types of laboratory quality-control samples: blanks, spikes, and replicates. Blanks and spikes were used to estimate result bias. They were prepared at the laboratory by using certified contaminant-free water or extraction reagents and processed them identically with the tissue extracts. Replicates were taken from the environmental tissue extracts and were used to estimate result variability. Additionally, surrogate compounds were added to each environmental sample at the laboratory to monitor the sample-specific performance of the analytical methods. Only four compounds had blank sample detections: BDE 100, 47, and 99; and triclosan. The spiked laboratory samples for each compound had recovery percentages that ranged from 32.5 to 137.2 percent. Recovery percentages for the surrogate compounds PCB202-13C12, dibromooctafluorobiphenyl, and ddt-d8 (n=16 each) ranged from 46.9 to 154.2 percent, 42.3 to 136.7 percent, and 48.8 to 317.8 percent, respectively, and the mean recovery percentages for those compounds were 67.4, 58.6, and 99.0 percent, respectively. Replicate samples were prepared and analyzed in conjunction with tissues used in Nilsen and others (2014), where 30 percent of samples were run in replicate. The resulting relative standard deviations for detected compounds of a sample were 23 percent for pesticides, 13 percent for PCBs, and 17 percent for PBDEs. All quality-assurance/quality-control results are available in the associated data release (Nilsen and others, 2016).
Analysis of Results
In this report, the analysis of the data was performed by (1) calculating the total concentration of each chemical class by site and tissue type (where the total concentration was the sum of concentrations for all chemicals within the chemical class), (2) calculating the total concentration of each chemical class for each tissue type, and (3) calculating the total concentration of each chemical class for each site. This analysis will inform the overall understanding of whether and where chemicals are accumulating in white sturgeon and may be used to assist in the evaluation of human-health criteria. The median total concentrations for the chemical classes (based on the values for all tissue types) also were calculated. Statistical analyses beyond what was previously mentioned in this report were not conducted because of the small sample size.
Results
The sampling results showed that OC pesticides and OC degradation products, PBDE congeners, and PCB congeners typically were present in all tissues and at all sites sampled, but that only one industrial or personal care product (triclosan) was found at any of the sites (tables 3 and 4)1. The PCB congeners were the most prevalent of any of the chemical classes—they were present in 91 percent of the samples analyzed for that chemical class, and each congener was detected at least once. The two PCB congeners with the highest total concentrations were PCB 180 and 101, constituting 18 and 14 percent of the total concentration of all PCBs summed, respectively. PBDE congeners were present in 57 percent of the samples analyzed for that chemical class and four (BDE 47, 100, 153, 154) were detected in all samples, whereas three others (BDE 71, 85, 183) were not detected. Of the total concentrations of all PBDEs summed, BDE 47 and 100 accounted for 72 and 20 percent of the total concentration, respectively. OC pesticides were present in 48 percent of the samples analyzed for that chemical class and were not detected in six samples. The results also showed that the banned OC pesticides included in this analysis (chlordane, dieldrin, and the DDT degradation products) continue to persist in the environment and that, although many currently used OC pesticides were detected, their total concentrations were relatively low across all sites and tissue types compared to the DDT degradation products (table 5). The total concentrations of the DDT degradation products were much higher than for any of the other chemical classes across all sites and tissue types and, as a result, this chemical class had the highest median total concentration, followed by the PCB congeners, the PBDE congeners, and the OC pesticides (table 5).
From this point on, (1) all results, interpretation, and discussion are focused exclusively on the three chemical classes other than industrial or personal care products; and (2) non-DDT OC pesticides and their degradation products are referred to as simply “OC pesticides.”
Table 3.
Detections of organochlorine pesticides and degradation products, industrial and personal care products, polybrominated diphenyl ether congeners, and polychlorinated biphenyl congeners in tissues (brain, fillet, gonad, and liver) of white sturgeon (Acipenser transmontanus) at sites 1–3 in the Hanford Reach of the Columbia River near Hanford, Washington.[Chemicals: BDE, brominated diphenyl ether; DDD, dichlorodiphenyldichloroethane; DDE, dichlorodiphenyldichloroethylene; DDT, dichlorodiphenyltrichloroethane; PCB, polychlorinated biphenyl. Abbreviations: ND, chemical was not detected; x, chemical was detected]
Table 4.
Detections of organochlorine pesticides and degradation products, industrial and personal care products, polybrominated diphenyl ether congeners, and polychlorinated biphenyl congeners in tissues (brain, fillet, gonad, and liver) of white sturgeon (Acipenser transmontanus) at sites 4–5 in the Hanford Reach of the Columbia River near Hanford, Washington.[Chemicals: BDE, brominated diphenyl ether; DDD, dichlorodiphenyldichloroethane; DDE, dichlorodiphenyldichloroethylene; DDT, dichlorodiphenyltrichloroethane; PCB, polychlorinated biphenyl. Abbreviations: ND, chemical was not detected; x, chemical was detected]
Table 5.
Sum of measured concentrations of persistent chemicals across four types of tissues (brain, fillet, gonad, and liver) measured in white sturgeon (Acipenser transmontanus) at each study site in the Hanford Reach of the Columbia River near Hanford, Washington.[Total concentrations measured for chemical class: values are equal to the sum of measured concentrations across all four types of tissue. Type of white sturgeon tissue: liver tissues were not analyzed at sites 1 and 3. Industrial or personal care products: triclosan was the only chemical detected. Abbreviations: DDT, dichlorodiphenyltrichloroethane; NA, not applicable; ND, chemical was not detected; OC, organochlorine; PBDE, polybrominated diphenyl ether; PCB, polychlorinated biphenyl; µg/kg, microgram per kilogram, wet weight]
Overall, gonad tissue had the highest total concentration of all chemicals summed from every site (see “All chemicals” row in table 6), followed by brain, liver, and fillet, but this pattern was not consistent across sites. For example, brain tissue had the highest total concentration of all chemicals at site 4 and the second highest at all remaining sites, whereas gonad tissues had the second highest total concentration of all chemicals at site 4 (see “All chemicals” column in table 5). Fillet tissues generally had the lowest concentrations and fewest contaminants detected, which is consistent with prior studies (Hulstrom, 2011). Studies of several sturgeon species reported that fillet tissues had lower overall lipid content while also having the lowest contaminant burden of lipophilic compounds (Wan and others, 2007; Mierzykowski, 2010; Peng and others, 2012); therefore, fillet tissues are assumed to have less affinity for OCs and other lipophilic compounds.
Although the lack of liver samples at sites 1 and 3 potentially biased the results shown in tables 5 and 6, these summaries are still useful in discerning overall patterns. For example, the highest total concentration of all chemicals was found at site 3 and the second highest was found at site 1, even though there were no liver results available for either site.
Table 6.
Sum of concentrations of persistent chemicals, by type of tissue (brain, fillet, gonad, and liver) and by site, measured in white sturgeon (Acipenser transmontanus) in the Hanford Reach of the Columbia River near Hanford, Washington.[Chemical class: DDT, dichlorodiphenyltrichloroethane; OC, organochlorine; PBDE, polybrominated diphenyl ether; PCB, polychlorinated biphenyl. Total concentrations measured in white sturgeon tissue: values are equal to the sum of measured concentrations across all five sites. Livers: liver tissues were not analyzed at sites 1 and 3. Total concentrations measured at each site: values are equal to the sum of measured concentrations across all four types of tissue. Abbreviations: NA, not applicable; µg/kg, microgram per kilogram]
Discussion
Chemical Accumulation in Tissues
The results from this study showed that persistent chemicals of concern are bioaccumulating in white sturgeon residing in the Hanford Reach of the Columbia River and that the most prevalent of the chemicals measured were endocrine disruptors that can impact the reproductive health of the fish (Beckvar and Lotufo, 2011). The highest concentrations of these chemicals were measured within the gonad tissues of the white sturgeon sampled in this study. Highly lipophilic endocrine disrupting chemicals, such as OC pesticides, PBDEs, and PCBs, can transfer from contaminated tissues in adults to developing eggs and concentrate in lipid-rich gonads (Beckvar and Lotufo, 2011). Although the pathways that these chemicals take within white sturgeon are not fully understood, maternal transfer of pollutants might explain the high concentrations of persistent chemicals measured in the gonads of the fish sampled in this study and others. Additionally, white sturgeon is a long-lived and a late maturing species, which allows for a longer period of time for chemicals to accumulate and eventually transfer into developing eggs. Growth and reproductive challenges were observed in Columbia River white sturgeon studied by other researchers (Feist and others, 2005) and in white sturgeon sampled in the San Francisco Estuary (Gundersen and others, 2017) that had similar chemical exposures to those measured in this study. In both studies, elevated concentrations of DDE and PCBs were detected at high frequencies in the liver and gonads, with similar detections observed in this study. The high concentrations of DDE and PCBs were attributed in the previous studies as a likely cause for growth, reproductive, and feminization effects on the white sturgeon. Further assessments likely would help in understanding the toxic effects on resident fish like white sturgeon and would help differentiate between the effects of endocrine disruption and carcinogenesis from the bioaccumulation of persistent chemicals.
Comparison to Previous Studies
Although total concentrations are useful for assessing patterns in the results from this study, comparing the results to those from previous studies helps inform the overall understanding of the bioaccumulation of chemical compounds in white sturgeon. The maximum concentrations of DDE and total PCB congeners for the fillet tissues analyzed in this study were compared to the fillet results from two other studies that sampled white sturgeon from the Hanford Reach (EPA, 2002; Hulstrom, 2011). The results from this study also were compared to the results from studies on white sturgeon sampled in other reaches of the Columbia River (Foster and others, 2001; Feist and others, 2005; and Gundersen and others, 2008) and in the San Francisco Estuary (Gundersen and others, 2017) to get a more complete understanding of the relative contaminant burdens of these fish.
Although the total concentrations of DDE and PCB congeners in fillets measured in this study were lower than comparable results from the two previous studies of white sturgeon in the Hanford Reach (EPA, 2002; Hulstrom, 2011), the overall results generally were consistent with those from previous studies of white sturgeon collected from other reaches of the Columbia River and in the San Francisco Estuary (Foster and others, 2001; Feist and others, 2005; Gundersen and others, 2008, Gundersen and others, 2017). The maximum total DDE concentrations from the EPA (2002) and Hulstrom (2011) studies were 3.7 and 2.2 times greater, respectively, than the maximum total DDE concentration from this study; the mean total PCB concentration from the EPA (2002) study and the maximum total PCB concentration from the Hulstrom (2011) study were 2.4 and 8.2 times greater, respectively, than the maximum total concentration of PCB congeners from this study. Like this study, the data from white sturgeon in other reaches of the Columbia River and in the San Francisco Estuary showed that DDT degradation products were the most commonly detected chemicals in white sturgeon sampled and were present at higher concentrations than other types of chemicals detected within their respective studies. Persistent chemicals such as DDE, PCBs, and PBDEs also had high detection frequencies in the liver and gonad tissues of white sturgeon in the San Francisco Estuary, reinforcing previous findings that bioaccumulation of persistent chemicals in white sturgeon is an issue beyond the Columbia River Basin. However, distribution of some compounds within the tissues can vary among studies. For example, total DDT and PCB concentrations measured in the Columbia River study by Feist and others (2005) and the study in the San Francisco Estuary by Gundersen and others (2017) were higher in liver tissue than in gonad tissue, whereas, in this study, total concentrations of DDT degradation products and PCB congeners were much higher in gonad tissues. Although the lack of liver samples at sites 1 and 3 potentially biased the results, detections of DDT degradation products and PCB congeners were approximately 10 times and 3.5 times higher in gonad tissues than in liver tissues, respectively.
Comparison to Human Health Benchmarks
A select set of available results from this study were compared to the EPA human health screening values (SVs) (EPA, 2000) and Washington State Department of Health (WSDOH) screening levels (SLs) (WSDOH, 2012, 2017) to evaluate the potential human health risks from consuming white sturgeon from the Hanford Reach (table 7). The SVs and SLs are the acceptable concentrations of bioaccumulative chemicals in fish tissue relative to the human general/recreational population and subsistence/Tribal populations. The SVs and SLs for the latter group are lower (and therefore more protective) because people in that group are expected to consume fish at higher rates than the general/recreational population and generally use whole fish in their food preparations. The SVs and SLs are calculated by the carcinogenic effects, defined as health effects that manifests over an entire lifetime, and noncarcinogenic effects, defined as acute health effects over a relatively short period of time (EPA, 2000; WSDOH, 2012, 2017). The EPA SVs and WSDOH SLs were available for eight of the chemicals or chemical combinations that were analyzed in this study (tables 8 and 9). Of the industrial or personal care products analyzed for this study, only one product in that chemical class (triclosan) was detected, with no available SV or SL for that chemical; therefore, no industrial or personal care product was used for any comparison. The risk to the general/recreational population was determined by comparing the fillet tissue results from this study to the applicable SVs and SLs. In contrast, the risk to subsistence/Tribal populations was determined by comparing the mean results for all tissues to the applicable SVs and SLs, based on the assumption that a whole-fish value likely would be within the range between the minimum and maximum tissue concentrations, and close to the mean value. The minimum and maximum concentrations for each chemical and chemical combination also are presented in tables 8 and 9.
Table 7.
U.S. Environmental Protection Agency (EPA) human health screening values and Washington State Department of Health (WSDOH) screening levels for select target analytes used for comparison between population types (general/recreational and subsistence/Tribal) and carcinogen risk levels.[Chemical class: DDT, dichlorodiphenyltrichloroethane; PCB, polychlorinated biphenyl. Total Chlordane: concentration of cis- and trans-chlordane, cis- and trans-nonachlor, and oxychlordane. Abbreviations: NA, not applicable; µg/kg, microgram per kilogram]
Table 8.
Comparison of analytical results of persistent chemicals in tissues of white sturgeon in the Hanford Reach of the Columbia River near Hanford, Washington, with U.S. Environmental Protection Agency (EPA) screening values for seven target chemicals.[Bold values indicate that at least one screening value was exceeded. Total Chlordane: total concentration of cis- and trans-chlordane, cis- and trans-nonachlor, and oxychlordane. DDT degradation products: total concentration of DDE, DDD, and DDT. PCB congeners: total concentration of all PCB congeners listed in table 1. Abbreviations: DDD, dichlorodiphenyldichloroethane; DDE, dichlorodiphenyldichloroethylene; DDT, dichlorodiphenyltrichloroethane; ND, chemical was not detected; PCB, polychlorinated biphenyl; µg/kg, microgram per kilogram, wet weight]
Table 9.
Comparison of analytical results of persistent chemicals in tissues of white sturgeon in the Hanford Reach of the Columbia River near Hanford, Washington, with Washington State Department of Health (WSDOH) screening levels for six target chemicals.[Bold values indicate that at least one acceptable tissue level was exceeded. Total Chlordane: total concentration of cis- and trans-chlordane, cis- and trans-nonachlor, and oxychlordane. DDT degradation products: total concentration of DDE, DDD, and DDT. Abbreviations: PBDE, polybrominated diphenyl ether; DDD, dichlorodiphenyldichloroethane; DDE, dichlorodiphenyldichloroethylene; DDT, dichlorodiphenyltrichloroethane; ND, Chemical was not detected; PCB, polychlorinated biphenyl; µg/kg, microgram per kilogram, wet weight]
A comparison of the results from this study to the EPA’s SVs and the WSDOH SLs indicated that the general/recreation population and subsistence/Tribal populations have increased health risks associated with the consumption of white sturgeon from the Hanford Reach. In general, EPA carcinogen SVs (fig. 2) had more chemical detections above their respective chemical threshold values for the general/recreation and subsistence/Tribal consumers than for non-carcinogen values. No non-carcinogen values exceeded the WSDOH SL for three of the six chemicals (dieldrin, hexachlorobenzene, and total chlordane) for both the general/recreational and subsistence/Tribal consumers (fig. 3). However, SLs for total DDT, total PBDE, and total PCB had detections above the threshold value for both general/recreational and subsistence/Tribal consumers, with all total PBDE detections for subsistence/Tribal consumers above the SL value. For the general/recreation population, the total concentration in fish fillet exceeded the carcinogenic SV at four of five sites for DDT degradation products and at one site for PCB congeners. None of the non-carcinogen fillet SVs for the general/recreation population were exceeded, however, site 4 exceeded one SL for PCB congeners. The results from this study also indicated that the health risks are greater for subsistence/Tribal populations than for the general/recreation population. The mean concentration in all tissue types exceeded the subsistence/Tribal, noncarcinogenic SV and SL at all five sites for DDT degradation products and exceeded the subsistence/Tribal, noncarcinogenic SV and SL at all five sites for PCB congeners. The mean concentration in all tissue types also exceeded SLs at all five sites for PBDE congeners. Additionally, the mean concentration in all tissue types exceeded the subsistence/Tribal, carcinogenic SVs at all five sites for DDT degradation products, dieldrin, and PCB congeners. The mean concentration in all tissue types also exceeded the subsistence/Tribal carcinogenic SVs at three and four sites for total chlordane and hexachlorobenzene, respectively.
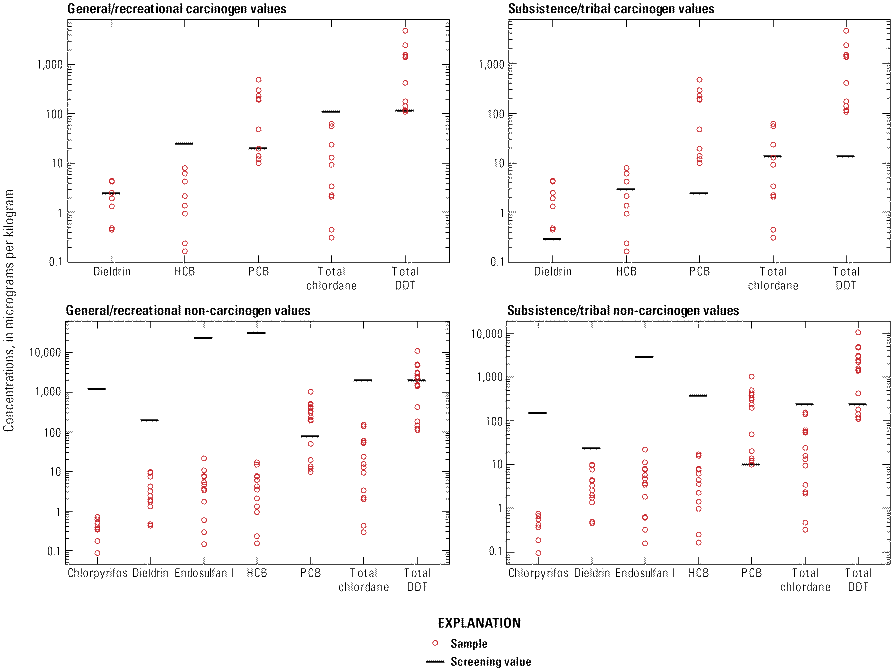
Chemicals detected in tissues of white sturgeon compared to U.S. Environmental Protection Agency screening values, in the Hanford Reach of the Columbia River near Hanford, Washington, 2009. [No chlorpyrifos and endosulfan I carcinogen screening values are available. DDT, dichlorodiphenyltrichloroethane; HCB, Hexachlorobenzene; PCB, polychlorinated biphenyl.]
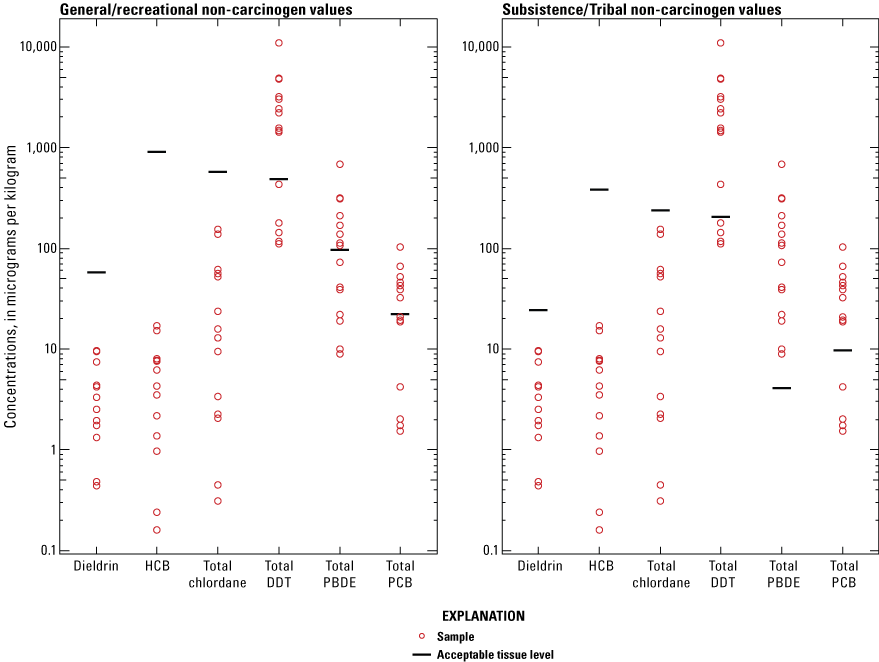
Chemicals detected in tissues of white sturgeon compared to Washington State Department of Health screening levels, in the Hanford Reach of the Columbia River near Hanford, Washington, 2009. [No carcinogen screening levels available. DDT, dichlorodiphenyltrichloroethane; HCB, Hexachlorobenzene; PBDE, polybrominated diphenyl ether; PCB, polychlorinated biphenyl.]
Conclusions
White sturgeon (Acipenser transmontanus) collected from the Hanford Reach of the Columbia River near Hanford, Washington, in 2009 had four tissue types analyzed for persistent chemicals of concern. This study sought to (1) determine whether persistent chemicals of concern are bioaccumulating in white sturgeon residing in the Hanford Reach, (2) identify where in the fish the chemicals are accumulating, and (3) determine whether there are implications for human health related to the bioaccumulation of persistent chemicals of concern in white sturgeon. Results showed that all chemical classes analyzed in this study accumulated in these fish, and, in general, each chemical class often was present in all tissues and at all sites within the study area. The most prevalent chemical classes detected were polychlorinated biphenyls, followed by polybrominated diphenyl ethers and organochlorine pesticides, respectively, although dichlorodiphenyltrichloroethane degradation products on their own had the highest concentrations detected of all chemical classes. Detection of these chemicals is consistent with previous studies of Columbia River white sturgeon and suggests that these highly persistent chemicals are frequently accumulating in white sturgeon and other aquatic species. Further examinations of prey items and substrate habitats would be useful in determining whether the chemical accumulation is occurring from the diet of the white sturgeon or is derived from elsewhere. Overall, the gonad tissue carried the most burden of the accumulated chemicals, having the highest concentrations detected of each chemical class. The detections of chemicals analyzed in this study highlight the importance of frequent monitoring of aquatic species used for human consumption. Comparisons to Environmental Protection Agency screening values and Washington State Department of Health screening levels showed that several threshold values were exceeded. Several agencies provide guidelines on the daily intake of fish to minimize the consumption of persistent chemicals of concern, and the results from this and other studies can play an important part in the decisions that go into developing those guidelines. Therefore, it would be useful for future studies to examine and continue to monitor chemicals bioaccumulating in resident fish to evaluate the potential health risk to consumers. Additionally, more extensive studies on the effects of accumulating chemicals in individual organs would help resource managers better understand the potential repercussions these chemicals have on the reproductive health and overall well-being of aquatic species.
References Cited
Agency for Toxic Substances and Disease Registry, 2011, Public health assessment Hanford Site, Richland, Benton County, Washington—EPA facility ID—WA3890090076: U.S. Department of Health and Human Services, 271 p., accessed June 15, 2020, at https://stacks.cdc.gov/view/cdc/21996.
Capron, J.M., 2008, Remaining sites verification package for the 128-F-2, 100-F burning pit waste, waste site reclassification form 2008-031: U.S. Department of Energy Report RSVP-2008-031, [variously paged], accessed February 10, 2020, at https://doi.org/10.2172/944172.
Feist, G.W., Webb, M.A.H., Gundersen, D.T., Foster, E.P., Schreck, C.B., Maule, A.G., and Fitzpatrick, M.S., 2005, Evidence of detrimental effects of environmental contaminants on growth and reproductive physiology of white sturgeon in impounded areas of the Columbia River: Environmental Health Perspectives, v. 113, no. 12, p. 1675–1682.
Foster, E.P., Fitzpatrick, M.S., Feist, G.W., Schreck, C.B., Yates, J., Spitsbergen, J.M., and Heidel, J.R., 2001, Plasma androgen correlation, EROD induction, reduced condition factor, and the occurrence of organochlorine pollutants in reproductively immature white sturgeon (Acipenser transmontanus) from the Columbia River, USA: Archives of Environmental Contamination and Toxicology, v. 41, p. 182–191.
Gundersen, D.T., Webb, M.A.H., Fink, A.K., Kushner, L.R., Feist, G.W., Fitzpatrick, M.S., Foster, E.P., and Schreck, C.B., 2008, Using blood plasma for monitoring organochlorine contaminants in juvenile white sturgeon, Acipenser transmontanus, from the Lower Columbia River: Bulletin of Environmental Contamination and Toxicology, v. 81, no. 3, p. 225–229.
Gundersen, D.T., Zeug, S.C., Bringolf, R.B., Merz, J., Jackson, Z., and Webb, M.A.H., 2017, Tissue contaminant burdens in San Francisco Estuary white sturgeon (Acipenser transmontanus)—Implications for population recovery: Archives of Environmental Contamination and Toxicology, v. 73, no. 2, p. 334–347, accessed June 15, 2020, at https://doi.org/10.1007/s00244-017-0378-9.
Hildebrand, L.R., Drauch Schreier, A., Lepla, K., McAdam, S.O., McLellan, J., Parsley, M.J., Paragamian, V.L., Young, S.P., 2016, Status of white sturgeon (Acipenser transmontanus Richardson, 1863) throughout the species range, threats to survival, and prognosis for the future: Journal of Applied Ichthyology, v. 32, no. S1, p. 261–312.
Hulstrom, L., 2011, Data summary report for the remedial investigation of Hanford Site releases to the Columbia River, Hanford Site: Washington: U.S. Department of Energy Report WCH-398, [variously paged], accessed February 20, 2020, at https://doi.org/10.2172/1084002.
Nilsen, E., Zaugg, S., Alvarez, D., Morace, J., Waite, I., Counihan, T., Hardiman, J., Torres, L., Patino, R., Mesa, M., and Grove, R., 2014, Contaminants of legacy and emerging concern in largescale suckers (Catostomus macrocheilus) and the foodweb in the lower Columbia River, Oregon and Washington, USA: The Science of the Total Environment, v. 484, p. 344–352.
Nilsen, E.B., Davis, J.W., and Hapke, W.B., 2016, Chemicals of emerging and legacy concern in tissues of white sturgeon (Acipenser transmontanus) in the Columbia River near Hanford, WA (2009): U.S. Geological Survey data release, accessed January 10, 2020, at https://doi.org/10.5066/F7Z899JC.
Pacific Northwest National Laboratory [PNNL], 2005, Survey of potential Hanford Site contaminants in the upper sediment for the reservoirs at McNary, John Day, The Dalles, and Bonneville Dams, 2003: Pacific Northwest National Laboratory Report DOH 320-034, PNNL 14878, WDOE 04-05-016, ODOE NUC-007, [variously paged].
Zaugg, S.D., Smith, S.G., Schroeder, M.P., Barber, L.B., and Burkhardt, M.R., 2007, Methods of analysis by the U.S. Geological Survey National Water Quality Laboratory—Determination of wastewater compounds by polystyrene-divinylbenzene solid-phase extraction and capillary-column gas chromatography/mass spectrometry: U.S. Geological Survey Water-Resources Investigations Report 2001–4186, 37 p.
Conversion Factors
Supplemental Information
Concentrations of chemical constituents in tissues are given in micrograms per kilogram (µg/kg), wet weight. No lipid content was provided.
Abbreviations
CRITFC
Columbia River Inter-Tribal Fish Commission
DDD
dichlorodiphenyldichloroethane
DDE
dichlorodiphenyldichloroethylene
DDT
dichlorodiphenyltrichloroethane
HCB
hexachlorobenzene
NWQL
National Water Quality Laboratory
ODEQ
Oregon Department of Environmental Quality
OC
organochlorine
PAH
polycyclic aromatic hydrocarbon
PBDE
polybrominated diphenyl ether
PCB
polychlorinated biphenyl
PNNL
Pacific Northwest National Laboratory
SL
screening level
SV
screening value
EPA
U.S. Environmental Protection Agency
USGS
U.S. Geological Survey
WSDOH
Washington State Department of Health
Publishing support provided by the U.S. Geological Survey
Science Publishing Network, Tacoma Publishing Service Center
For more information concerning the research in this report, contact the
Director, Oregon Water Science Center
U.S. Geological Survey
2130 SW 5th Avenue
Portland, Oregon 97201
Suggested Citation
Payne, S.E., Wise, D.R., Davis, J.W., and Nilsen, E.B., 2022, Assessment of persistent chemicals of concern in white sturgeon (Acipenser transmontanus) in the Hanford Reach of the Columbia River, southeastern Washington, 2009: U.S. Geological Survey Scientific Investigations Report 2022–5020, 25 p., https://doi.org/10.3133/sir20225020.
ISSN: 2328-0328 (online)
Study Area
| Publication type | Report |
|---|---|
| Publication Subtype | USGS Numbered Series |
| Title | Assessment of persistent chemicals of concern in white sturgeon (Acipenser transmontanus) in the Hanford Reach of the Columbia River, southeastern Washington, 2009 |
| Series title | Scientific Investigations Report |
| Series number | 2022-5020 |
| DOI | 10.3133/sir20225020 |
| Year Published | 2022 |
| Language | English |
| Publisher | U.S. Geological Survey |
| Publisher location | Reston, VA |
| Contributing office(s) | Oregon Water Science Center |
| Description | vii, 25 p. |
| Country | United States |
| State | Washington |
| Other Geospatial | Columbia River |
| Online Only (Y/N) | Y |
| Google Analytic Metrics | Metrics page |


